Limitations
Really cool paper out in JACS yesterday detailing an electrophilic aromatic substitution reaction that works on really electron-deficient rings. With some cheap pool chemicals, a pretty simple benzenesulfonic acid catalyst, and the world's most magical solvent, you can stick a bromine on some pretty tough rings.
But as interesting as that is, my favorite part of the paper is the portions of the figures that list the method's limitations.
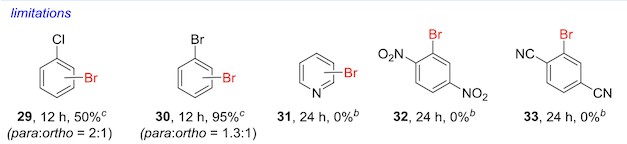
There's an obvious incentive in a methods paper like this to market the method in as positive a light as possible. A huge percentage of papers probably leave out results that didn't turn out well because it makes the method look weaker. But when you do see this sort of thing, it's on really high-quality papers – and it's pretty likely that you can trust the method to work on the first shot.